|
I have naturally started tinkering with these emulsions (by assigning a student to studying this type of emulsions). Oil-in-water Pickering emulsion refers to an emulsion where droplets of oil are dispersed in water as the continuous phase. I find these emulsions interesting, since it is not completely clear to me what physical parameters are responsible for the apparent meta-stability of these emulsions. Proteins, polysaccharides, and their composite particles can be used as good particle. These types of emulsions are known as “Pickering” emulsions and are a type of particle super-assembly. Pickering emulsion, which is an emulsion stabilized by solid particles, offers a wide range of potential applications because it generally provides a more. High internal phase Pickering emulsion (HIPPE) is a type of emulsion stabilized by solid particles irreversibly adsorbed on an interfacial film, and the volume fraction of the dispersed phase () is larger than the maximum packing volume fraction ( max). When you shake oil and vinegar together (with a speck of mustard) you form a semi-stable vinaigrette, which is stabilized by small insoluble mustard particles. The milk-fat droplets in homogenized milk are stabilized by small protein-covered calcium particles that adsorb to the fat/whey interface. Particle stabilized emulsions are found in many (tasty) applications. Can we now manipulate these particles and have them assemble into larger particles? This is a form of nano-assembly, which is controlled by a mini-emulsion template. So, we make polymer nano-capsules by enclosing a 100 nm droplet of an oil solution within a 10 nm thick polymer shell. Here, we review recent findings concerning Pickering emulsions, with a particular focus on how the nanoparticles morphology (i.e., cube, ellipsoid, nanosheet, sphere, cylinder, rod, peanut) influences the type and stability of such emulsions, and their current applications in different fields such as antibacterial activity, protein recognition, catalysis, photocatalysis, and water purification.HIERARCHICAL ASSEMBLY OF POLYMER CAPSULES Moreover, the nanoparticle morphology strongly influences Pickering emulsion stability as well as the potential utilization of such emulsions. Therefore, they can be considered attractive components for various uses, such as photocatalysis and the preparation of new materials. Furthermore, Pickering emulsions show higher stability, lower toxicity, and stimuli-responsiveness, compared with emulsions that are stabilized by surfactants. In the past 10 years a new application has emerged in the field of catalysis to use them as vehicles to carry out catalytic reactions, allowing a more environmentally friendly process with high conversions and selectivities and important advantages for catalyst recovery. For emulsions with a higher concentration of nanoparticles, a larger number of polymeric nanoparticles could be used to cover the oilwater interface and then stabilize the emulsions. S2, as the concentration of micellar aggregates increased, the droplet size of the Pickering emulsion decreased. In contrast to classical emulsions, in Pickering emulsions, solid microparticles or nanoparticles that localize at the interface between liquids are used as stabilizers, instead of surfactants, to enhance the droplet lifetime. Particle-stabilised or Pickering emulsions are versatile systems. The Pickering emulsions were prepared by homogenizing a mixture of toluene and deionized (DI) water at 15 000 rpm for 120 s with different emulsifier concentrations at pH 2. 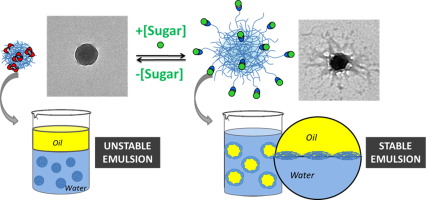
In recent years, Pickering emulsions and their applications have attracted a great deal of attention due to their special features, which include easy preparation and enhanced stability.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
